On July 8, in the wake of the Federal Trade Commission’s (FTC) designation of July as “Made in the USA” (MUSA) Month, the FTC issued warning letters to four companies suspected of violating the FTC’s Made in USA Labeling Rule under 16 C.F.R. § 323 (MUSA Labeling Rule) for deceptively claiming their consumer goods are of U.S.-origin and sent additional letters to online retailers, highlighting suspected similar violations by third-party sellers on their platforms. This move signals the FTC’s commitment to prioritizing MUSA labeling compliance and suggests that online retailers may need to take proactive measures to address false labeling and U.S.-origin claims on their platforms.
The FTC enforces both the FTC Act and the MUSA Labeling Rule, which address different aspects of product origin representations. The FTC Act prohibits unfair or deceptive acts in commerce, requiring that any representations, including origin claims, be truthful, non-misleading, and adequately substantiated. Meanwhile, the MUSA Labeling Rule governs labeling practices, stipulating that a product can only be labeled as “Made in the United States” if it meets certain criteria. To claim U.S. origin and label a product “Made in the United States,” a company must establish that a product is “all or virtually all made in the U.S., which means “the final assembly or processing of the product occurs in the United States, all significant processing that goes into the product occurs in the United States, and all or virtually all ingredients or components of the product are made and sourced in the United States.”
In response to “information and complaints” from unidentified parties, the FTC sent warning letters to four companies regarding their suspected violations of the FTC Act and MUSA Labeling Rules. These warning letters signaled that the companies could be subject to enforcement actions if they cannot adequately substantiate that the products meet the “all or virtually all” standard. Similarly, the FTC sent letters to two major online retailers, informing them of the MUSA labeling and advertising laws and how the FTC suspects that third-party sellers on their platforms are violating the “all or virtually all” standard. The FTC also signaled that online retailers should take corrective action “against third-party sellers who make false or misleading ‘Made in USA’ claims on [their] online marketplace in violation of the FTC Act, MUSA Labeling Rule, and [their seller codes of conduct].”
The FTC’s actions, including the declaration of July as MUSA Month, the issuance of warning letters to individual companies, and the advisory letters to leading online retailers, underscore its prioritization of enforcement of the MUSA Labeling Rule. Businesses must ensure their products, and those of third-party sellers, meet MUSA requirements to avoid potential penalties.
Nick Erickson, a 2025 summer associate with Troutman Pepper Locke who is not admitted to practice law in any jurisdiction, also contributed to this article.
This article was republished in IP Litigator on September 1, 2025.
I. Introduction
The evolution of grid management strategies to accommodate the increased adoption of renewable energy sources has led to a significant rise in both patent filings and patent disputes. An increased proportion of a grid’s power originating from variable energy sources, e.g., wind and solar, whose generation capacity fluctuates with weather, can pose challenges to grid stability and reliability. For example, intermittent energy sources like wind and solar can create “dips” in grid capacity that must be filled if demand simultaneously peaks. Issues include maintaining frequency and voltage stability as generation increases or decreases with conditions, leading to reduced grid inertia — the ability of an electrical power grid to resist changes in frequency due to sudden imbalances between electricity supply and demand. As a result, the grid management and control systems that balance supply and demand must be increasingly sophisticated to accommodate the difficulties in grid regulation.
The widespread power outages in Spain and Portugal during the spring of 2025 highlight concerns with energy grids, both in the U.S. and abroad, that increasingly incorporate renewable energy sources like solar and wind. While some blame Spain’s blackouts on the penetration of renewable energy sources, which has grown in recent years — from 43% in the 2010s to 56% in 2024[1] — others argue that grid management is to blame.[2] Rather than place the blame wholly on renewable energy penetration, these commenters suggest the more nuanced position that “investments in power storage and grid upgrades must go hand in hand with the expansion of renewables generation.” Increased investment in patent filings goes hand in hand with 21st-century grid upgrades, and recent litigation case studies demonstrate that patent disputes naturally follow increases in patent investment.
The core business models of energy producers and providers are traditionally not associated with significant IP ownership disputes and litigation risk. However, these core business models are facing new challenges related to IP while integrating new technologies. Two exemplary case studies of patent litigation illustrate these new challenges related to emerging renewable energy technologies. The cases relate to solar-related grid technologies and electrical energy storage. Each of these technologies contributes to overall grid management strategies; solar-related grid technologies aim to locally manage individual solar modules’ variable energy generation, and electrical energy storage technologies improve grid inertia by fulfilling demand when power generation from variable sources is limited. Innovation in these technologies is creating a complex IP landscape, fraught with potential litigation risks. Companies in these industries should take steps to proactively mitigate legal exposure.
II. Case Study: Solar-Related Grid Technologies
Residential solar panels allow homeowners to reduce their electric bill both by reducing their reliance on the grid and by selling excess power back to the grid. But with increased residential solar, grid management becomes exponentially more complex — rather than managing one central powerplant, every solar-generating house becomes a potential power source.
A. IP Landscape
The increasing complexity of residential power grids has generated a boom in patent filings covering technologies related to grid management. For example, patent filings on solar-related grid technologies, like grid-tied photovoltaic systems, backup power control, and load leveling functions have increased substantially in the past 25 years. The resulting patent landscape creates infringement risk for companies operating in this space and highlights the need for these companies to carve out their own IP protections.
Fig. 1: Yearly average patent publications filed under CPC codes for solar related grid technologies, per five-year period from 2000-2025. Compiled from USPTO patent database.[3]
B. Litigation Example
In one recent case, Relink US LLC sued a supplier of residential solar inverters over alleged infringement of Relink’s U.S. Patent No. 9,793,755. Originally filed in 2023, the case was stayed pending an inter partes review by the USPTO and had that stay lifted in April 2025.[4] At issue is Relink’s ‘755 patent, directed to an uninterruptible power supply (UPS) and method for managing grid-tied solar system.[5] The patent describes control of a solar system UPS with grid-tie, backup power control, and load-leveling functions.[6] As residential solar becomes increasingly prevalent, localized solutions are necessary to manage the needs of the complex grid. For example, load-leveling technology — which is described in the ‘755 Patent[7] — balances the supply and demand of power by storing produced energy when demand is low and distributing stored energy back to the grid when demand is high. For example, the ‘755 states:
The proliferation of distributed power generation has caused some problems in maintaining the stability of the utility grid. As a result, many electric utilities world-wide are seeking increased functionality in grid-tied inverters to help stabilize the grid. For example, some require grid-tied photovoltaic inverters to gradually reduce output power when the grid frequency deviates beyond a set limit[8]
The patent goes on to detail control strategies for improving grid stability, including measuring grid frequency and using string inverters to limit power output when grid frequency increases.[9]
Technologies like load leveling are critical to managing a complex, 21st-century grid, and these new technologies will be added to new products and utilized in new grid management techniques. While the Relink case is ongoing, it further demonstrates the IP risks on both sides for companies introducing and utilizing new technologies in the grid-management and residential solar industries.
III. Case Study: Electrical Energy Storage
Electrical energy storage technologies are becoming increasingly vital as the world transitions to renewable energy sources. These technologies, which include batteries, supercapacitors, and other storage systems, enable the efficient capture and use of energy generated from intermittent sources like solar and wind. As the demand for reliable and sustainable energy storage solutions grows, so too does the demand for investment and innovation in this field.
A. IP Landscape
This surge in technological advancements has led to a significant increase in the number of patents filed related to electrical energy storage technologies, covering various aspects such as materials, design, and methods of energy storage and retrieval. The rapid growth in patent filings has created a crowded IP landscape in the electrical energy storage sector. Companies looking to innovate must navigate a dense patent thicket, which can be a daunting and risky endeavor. The likelihood of inadvertently infringing on another company’s patent may be high, given the extensive and diverse range of patented technologies.
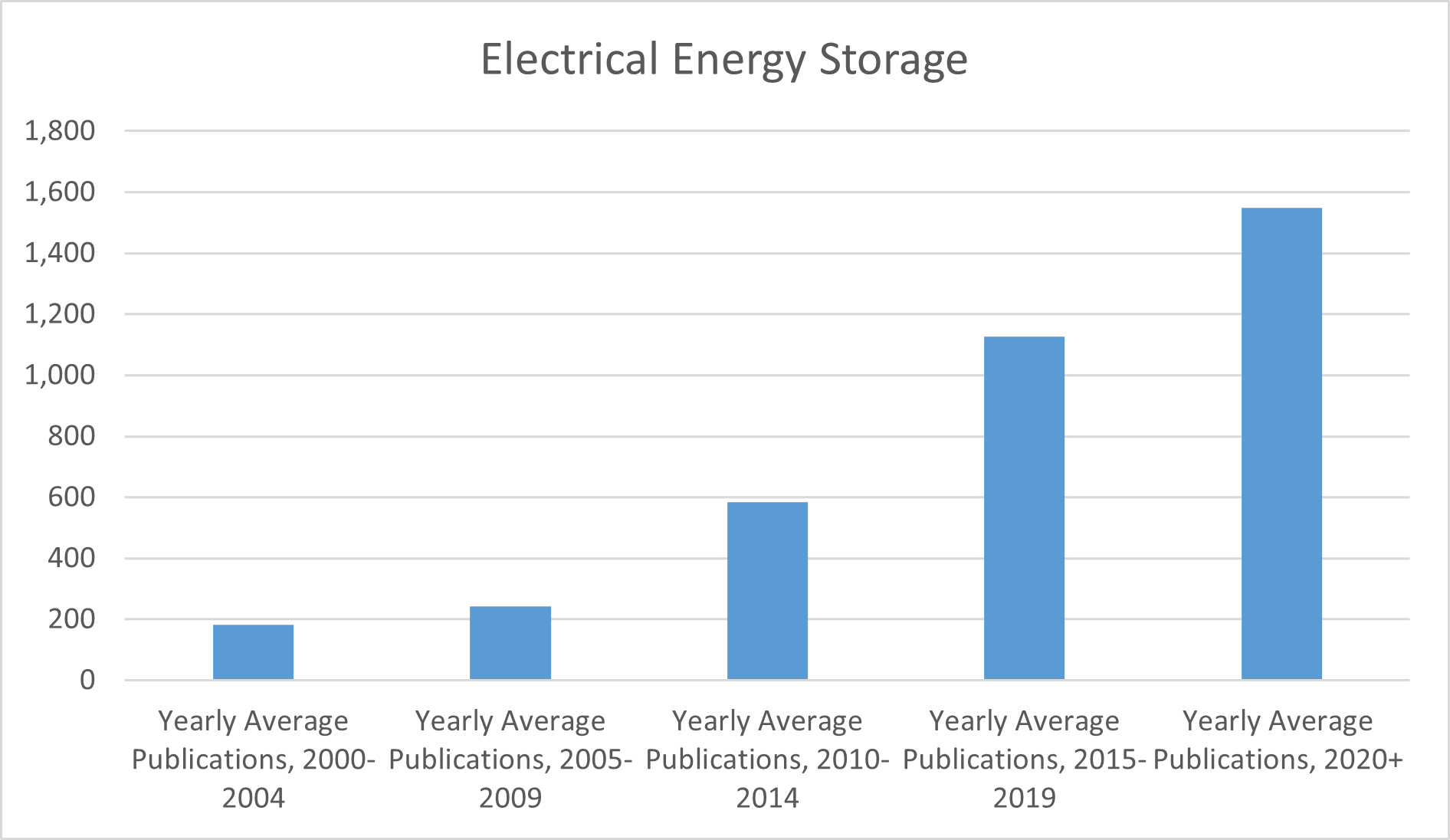
Fig. 2: Yearly average patent publications filed under CPC codes related to electrical energy storage technologies, per five-year period from 2000-2025. Compiled from USPTO patent database.[10]
B. Litigation Example
Two cases highlight the litigation risks associated with operating in this technology and IP-dense environment. In the first, a major lithium-ion battery manufacturer filed a $1 billion lawsuit against its supplier, accusing the company of misappropriating trade secrets related to electric vehicle battery manufacturing and incorporating them into patent filings.[11] Similarly, SK Innovation reached a settlement with LG Energy Solutions to avoid a U.S. import ban on its electric vehicle batteries, agreeing to pay LG Energy Solutions $1.8 billion.[12] While both those cases involve electric vehicle battery technology, the increase in patent infringement flings is not so limited. For example, LG Energy Solution recently alleged an increase in infringement of its battery-related patent portfolio and committed to taking “strict measures against unlawful infringements.”[13] These cases highlight the acceleration of both innovation and litigation in the battery sector and the high monetary value of IP assets.
IV. Mitigating Risks
To mitigate the risks associated with potential IP disputes, companies operating in renewable energy and grid-management industries should consider conducting thorough patent searches and seek expert legal advice. Identifying potential patent conflicts early in the development process can help companies avoid costly litigation and focus on innovation.
Additionally, companies should consider patenting their own technological advancements to protect their IP and establish a competitive edge. By building a robust patent portfolio, companies can create opportunities for cross-licensing and strategic partnerships, which can be beneficial in navigating the complex patent landscape.
V. Conclusion
The investment and IP growth associated with the surge in renewable energy penetration and increased grid complexity presents significant opportunities as well as risks. Companies can manage these risks through comprehensive patent searches, consulting legal counsel, and strategic patenting of their innovations. By doing so, they can safeguard their investments and contribute to the advancement of these technologies. As the industry continues to evolve, proactive management of patent exposure will be crucial for companies aiming to succeed in these promising yet challenging fields.
[1] Spain’s grid denies dependence on solar power to blame for blackout, Reuters, April 30, 2025 (available at https://www.reuters.com/world/europe/sanchez-pressed-explain-spains-blackout-grid-says-solar-not-blame-2025-04-30/#:~:text=The%20share%20of%20renewables%20as,is%20targeting%2081%25%20by%202030.).
[2] Don’t blame renewables for Spain’s power outage, Reuters, April 30, 2025 (available at https://www.reuters.com/business/energy/dont-blame-renewables-spains-power-outage-bousso-2025-04-30/).
[3] This data for this graph was generated using the USPTO’s Patent Public Search website (https://ppubs.uspto.gov/pubwebapp/). A set of CPC codes were selected based on their inclusion in WIPO’s “IPC Green Inventory” (https://www.wipo.int/classifications/ipc/green-inventory/home) and relation to solar-related power supply and grid management devices and methods.
[4] W.D. Tx. Case No. 1:23-cv-1093-DAE, Order (1) Lifting Stay of Case; (2) Granting Motion to Transfer Venue (April 3, 2025); after transfer, N.D. Ca. Case No. 3:25-cv-03365-RFL.
[5] U.S. Patent No. 9,793,755.
[6] Id.
[7] ‘755 Patent at 2:58-65 (“The controller may compare a net power being consumed by the load and produced by the current-source inverters to an allowable peak load power. If the net power is lower than the allowable peak load power, the controller may direct power from the AC voltage source to the battery. If the net power is higher than the allowable peak load power, the controller may direct power from the battery into the AC voltage source.”).
[8] ‘755 Patent at 1:24-30.
[9] ‘755 Patent at 6:28-34 (“the system may employ string inverters or substantially any other current-source inverters designed to limit power when the frequency of the grid 100 varies by a predetermined amount from the nominal grid frequency. For example, the current-source inverters may be designed to linearly decrease their power output from 100% to 40% when the frequency increases from 50.2 Hz to 50.5 Hz.”).
[10] This data for this graph was generated using the USPTO’s Patent Public Search website (https://ppubs.uspto.gov/pubwebapp/). A set of CPC codes were selected based on their inclusion in WIPO’s “IPC Green Inventory” (https://www.wipo.int/classifications/ipc/green-inventory/home) and relation to electrical energy storage systems and methods.
[11] https://www.law360.com/articles/1848668/tesla-slaps-supplier-with-1b-ev-battery-trade-secrets-suit.
[12] https://www.bloomberg.com/news/articles/2021-04-10/ford-vw-battery-supplier-said-to-reach-deal-to-avoid-import-ban.
[13] https://news.lgensol.com/company-news/press-releases/2636/.
On May 27, the Federal Communications Commission (FCC) opened two rulemaking proceedings aimed at foreign involvement in U.S. communications networks, particularly by countries identified as foreign adversaries, such as China and Russia. First, a Notice of Proposed Rulemaking (NPRM), titled “Protecting our Communications Networks by Promoting Transparency Regarding Foreign Adversary Control,” aims to expand foreign adversary ownership and control reporting requirements for FCC licensees. Second, a Report and Order and Further Notice of Proposed Rulemaking (FNPRM), titled “Promoting the Integrity and Security of Telecommunications Certifications Bodies, Measurement Facilities, and the Equipment Authorization Program,” finalizes the prohibition on certification, accreditation, or testing of equipment requiring FCC authorization by entities that are “owned by, controlled by, or subject to the jurisdiction or direction of” prohibited entities, and requests comment on expansion of equipment authorization program prohibitions and promotion of testing and certification within the United States.
The FCC is accepting comments on the NPRM until July 21 and reply comments by August 19. Comments on the FNPRM are due August 15, and reply comments are due September 15.
NPRM – Expansion of Foreign Adversary Ownership/Control Reporting Requirements
The rules proposed in the NPRM seek to expand the foreign adversary ownership and control reporting requirements by requiring entities holding a “Covered Authorization” to certify whether they are “owned by, controlled by, or subject to the jurisdiction or direction of a foreign adversary.” Those listed as foreign adversaries are China (including Hong Kong and Macau), Cuba, Iran, North Korea, Russia, and the Maduro Regime in Venezuela pursuant to 15 CFR § 791.4 . “Covered Authorization” is defined broadly to include holders of licenses, authorizations or approvals for wireless, satellite, media, submarine cable, telephone, and common carrier operations. It would also include entities seeking equipment certifications; holders of Telecommunications Relay Services and Data Network Identification Codes; and participants in FCC auctions.
This new reporting regime would significantly expand existing foreign ownership disclosure requirements that apply to a subset of entities, such as parties whose transactions — international telecommunication authorizations (Section 214), submarine cable authorization from a U.S. point to an international territory, or Section 310(b) requests for wireless and broadcast carriers to exceed the statutory foreign ownership limit — may implicate Team Telecom review.
Each “Covered Authorization” holder would be required to certify whether “it is or is not owned by, controlled by, or subject to the jurisdiction or direction of a foreign adversary.” If the entity answers in the affirmative, then it would have to disclose the following information:
-
All 5% or greater direct or indirect ownership interests (equity and/or voting interest);
-
Any natural persons with a 5% or greater interest will have to disclose their countries of citizenship.
-
Business organizations with a 5% or greater interest will need to disclose places of incorporation/organization, headquarters, and/or principal place of business.
-
-
The foreign adversary that the Covered Authorization holder is “owned by, controlled by, or subject to the jurisdiction or direction of”; and
-
The nature of the foreign adversary’s “ownership, control, jurisdiction, or direction” with respect to the Covered Authorization holder.
This reporting structure would reduce the threshold of existing foreign ownership reporting for certain licensees that are currently required to report 10% or greater foreign ownership, such as those seeking licenses through the competitive bidding process in wireless services under 47 CFR § 1.2112 or Section 214 transfer of control requests for international and domestic common carriers under 47 CFR Part 6.
The NPRM proposes to require annual certification from all Covered Authorization holders or file an initial certification with the condition that Covered Authorization holders report, within 30 days, any foreign ownership changes where a foreign adversary is involved or if there is a new foreign entity owning 5% or more.
For Covered Authorization holders that fail to respond or provide false certification information, the FCC is proposing to adopt a streamlined revocation process that requires notice to the Covered Authorization holder of the Commission’s intent to revoke its authorization with an opportunity to show cause why the authorization should not be revoked. The NPRM also considers whether Covered Authorization holders that report foreign ownership should be automatically referred to Team Telecom for a national security review — an uncommon practice since only certain applicants and licensee holders are subject to Team Telecom review.
Report and Order and FNPRM – Prohibition on Ownership/Control of Certification and Testing Bodies by Foreign Adversaries and Designated Entities
Under the Report and Order and FNPRM, the FCC will require Telecommunication Certification Bodies (TCB), test labs, or laboratory accreditation bodies (“testing entities”) to certify that they are not “owned by, controlled by, or subject to the jurisdiction or direction of a prohibited entity,” which is defined to include those described on the following lists:
-
FCC Covered List pursuant to Section 2 of the Secure and Trusted Communications Networks Act (STCNA);
-
Department of Commerce, Bureau of Industry (BIS) Entity List;
-
Department of Homeland Security Uyghur Forced Labor Prevention Act Entity List;
-
Section 5949 of the 2023 National Defense Authorization Act List of Semiconductor Companies including Semiconductor Manufacturing International Corporation (SMIC), ChangXin Memory Technologies (CXMT), Yangtze Memory Technologies (YMTC), and their affiliates;
-
Department of Defense 1260H List of Chinese Military Companies;
-
Department of the Treasury, Office of Foreign Assets Control (OFAC) List of Chinese Military-Industrial Complex Companies (NS-CMIC); and
-
Foreign Adversaries identified by Commerce under 15 CFR § 791.4, as listed above.
As part of its equipment authorization process, which is intended to regulate harmful interference by products that emit radiofrequency (RF) energy, the FCC delegates the testing and certification process to approved testing entities. According to this new rule, a testing entity cannot be “owned by, controlled by, or subject to the direction of a prohibited entity,” based on the lists above. Testing entities found to fit this definition will lose their recognition in the equipment authorization program and the FCC will “prohibit reliance on or use of, for purposes of equipment authorization, any [testing entity] owned by, controlled by, or subject to the direction of a prohibited entity.”
Under the FCC’s new rules, each recognized TCB, test lab, and laboratory accreditation body must certify to the FCC that it is not owned or controlled by, or subject to the direction of, a prohibited entity. These certifications would be due 30 days after the effective date of the rules. Then, within 90 days after the effective date of the rules, each TCB, test lab, and laboratory accreditation body must disclose to the Commission all equity or voting interests of 5% or greater.
If there are any ownership or control changes, testing entities will have 30 days to update their information.
Proposed Rules
The FNPRM also initiates a public comment process for several proposed actions related to this rule. Some notable proposed actions include the following:
-
Incorporation of additional federal lists to identify prohibited entities (e.g., Protecting Americans from Foreign Adversary Controlled Applications Act (PAFACA), OFAC Specially Designated Nationals and Blocked Persons List or 2024 National Defense Authorization Act (NDAA) list of prohibited battery manufacturers and its successors);
-
Adoption of a “presumption-of-prohibition policy” where an entity must provide clear and convincing evidence that there is no national security risk from its participation in the equipment authorization program; and
-
Rule changes to incentivize (such as expedited processing) the testing and certification of equipment by entities based in the U.S. or partner countries.
Moving Forward
These recent actions signal the FCC’s continuing shift to focus more on national security issues. Another illustration of this trend is Chairman Brendan Carr’s recent establishment of an FCC Council on National Security, with the following objectives:
-
Reduce the American technology and telecommunications sectors’ trade and supply chain dependencies on foreign adversaries;
-
Mitigate America’s vulnerabilities to cyberattacks, espionage, and surveillance by foreign adversaries; and
-
Ensure the U.S. wins the strategic competition with China over critical technologies, such as 5G and 6G, AI, satellites and space, quantum computing, robotics and autonomous systems, and the Internet of Things.
Recently, the FCC’s Bureau of Public Safety and Homeland Security sought expedited notice and comment (15-day response) on including in its Covered List certain hardware and software related to connected vehicles that the Commerce Department found to pose a national security risk under the ICTS regulations.
As the comment deadlines near, potentially impacted organizations should consider engaging in the process. More broadly, it is becoming increasingly clear that those in the FCC ecosystem will need to pay closer attention to potential national security issues as a condition of doing business in the United States. Entities that undertake offshore equipment authorization should consider how these rules and proposals may affect their operations. Telecom licensees and other entities holding “Covered Authorizations” should likewise consider any foreign ownership reporting requirements and establish a protocol for managing any new applicable requirements.
Troutman Pepper Locke’s telecommunications and national security teams will continue to monitor any updates related to these actions that could affect the telecommunication industry, and can assist relevant entities engage with regulators on these issues or implement processes to ensure regulatory compliance.
On July 4, 2025, President Trump signed the “One Big Beautiful Bill Act” (OBBB), H.R. 1, into law. The OBBB extended and made permanent the general qualified opportunity fund (QOF) legislative framework for investments in qualified opportunity zones (QOZs), while modifying certain significant aspects of the law, including an increased focus on investments in rural areas. This article offers a brief summary of the existing law, enacted by the Tax Cuts and Jobs Act (TCJA) in late 2017, and highlights the significant changes made by the OBBB.
Existing Law
QOZs are designated low-income census tracts where investments can receive preferential tax treatment if made through a QOF that invests at least 90% of its assets in qualified opportunity zone property (very generally, “original use” or “substantially improved” property located in a QOZ or stock or partnership interests in an entity operating a business and owning property in a QOZ). Taxpayers could defer eligible capital gains until December 31, 2026, by investing them in a QOF, generally within 180 days of the gain’s recognition, with the potential for partial exclusion of the deferred gain if the investment was held for at least five (10% exclusion of invested gain) or seven years (additional 5% exclusion). The five-year and seven-year holding periods had to be met prior to December 31, 2026, so this benefit has not been available for several years. An investor in a QOF can obtain a full exclusion of post-investment appreciation if it holds its interest in the QOF for at least 10 years.
Changes Under the OBBB
While the OBBB retains the general framework associated with investments in QOFs (e.g., the type of gain that can be invested in an QOF, general requirements for QOF, and qualified opportunity zone business (QOZB) qualification), the most notable changes introduced by the OBBB are highlighted below:
-
QOZs Made Permanent With “Rolling” 10-Year Designations. The OBBB permanently renews the QOZ program and introduces new, rolling 10-year periods for QOZ designations, with governors designating new QOZs every ten years beginning July 1, 2026 (Decennial Determination Dates) and with the first new designations taking effect January 1, 2027.
-
More Stringent QOZ Designation Criteria. The criteria for census tracts that are eligible to be designated as QOZs has been narrowed, reducing the median family income requirements, and the contiguous tract rule (allowing looser requirements for certain census tract contiguous to other qualifying tracts) has been eliminated.
-
Modified Gain Exclusion. Unlike the TCJA QOF rules, which allowed deferral until a fixed date such that the timing of investment determined whether the five-year (10%) or seven-year (additional 5%) gain exclusion rules (or no gain exclusion at all) would apply, the OBBB always allows a five-year deferral (assuming no intervening gain “inclusion events” take place) and at the end of holding the investment for five years—and prior to inclusion being triggered — there is a 10% gain exclusion. While the five-year deferral is less than the deferral that would have been available for an early investment under the old TJCA law (and there is no possibility of an additional 5% exclusion), the OBBB ensures some meaningful level of deferral regardless of the timing of an investment. The exclusion for post-investment appreciation for QOF investments held for more than 10 years still applies.
-
Enhanced Benefits for Investments in Rural QOZs. If a QOF qualifies as a Qualified Rural Opportunity Fund, which generally occurs if 90% or more of its property is located in a rural QOZ, the 10% gain exclusion after a five-year hold is increased to 30%. Additionally, the “substantial improvement” requirement to make property held by the QOF or QOZB good “qualified opportunity zone business property” is reduced, requiring an additional investment in such property equal to only 50% of the initial investment in the property (rather than 100%).
-
Increased Reporting Requirements. In an effort to increase transparency and the government’s ability to evaluate the effectiveness of the QOF legislation, QOFs and QOZBs will be required to supply additional information to the government regarding the value of their property and information about employees and investors. Penalties apply for failure to comply.
Future guidance from Treasury is expected to provide additional details on the new legislation, including its interaction with existing QOF legislation. Please feel free to reach out if you have any questions about how the revised QOF legislation will affect your existing investments in QOFs and future fund and real estate investments.
This article was originally published on July 18, 2025 in Reuters and Westlaw Today.
Intoxicating hemp-derived products have proliferated across the U.S. under the guise of the 2018 Farm Bill’s definition of “hemp.” Although these products produce psychoactive effects akin to state-regulated cannabis products, they are often manufactured and sold with little oversight or regulatory controls.
Lawmakers and regulators have intensified efforts to address the Farm Bill’s perceived loophole, prompting new legislation, regulatory action, and litigation. Manufacturers and retailers argue that the Farm Bill preempts state regulatory efforts to restrict these products. This article outlines significant federal and state developments related to the regulation of intoxicating hemp-derived products in the U.S. over the last year.
Background: The Delta-9 THC Loophole
The 2018 Farm Bill aimed to legalize the cultivation, manufacture, and distribution of industrial hemp products by carving “hemp” out of the definition of “marijuana” under the federal Controlled Substances Act. Congress did so by defining hemp as any part of the cannabis plant which contains less than 0.3% delta-9 THC on a dry weight basis. At the time, delta-9 THC was the most well-known psychoactive compound of the cannabis plant.
Shortly thereafter, innovative market actors developed methods to exploit less well-known cannabinoids that also have psychoactive effects — such as delta-8 and delta-10 THC — and create intoxicating products that technically fall below the delta-9 THC threshold. This has allowed manufacturers and retailers to develop and sell these products without regulatory oversight, all while claiming that they are federally legal.
The Food and Drug Administration maintains that it cannot regulate intoxicating cannabinoids under its existing legal authority and has urged Congress to act. While some attempts have been made to address the Farm Bill’s loophole through legislation, those efforts have, so far, been unsuccessful.
Federal Legislative Proposals
In June 2025, the U.S. House Appropriations Committee advanced a spending bill with provisions designed to redefine “hemp” and ban most THC-containing hemp products. Championed by Rep. Andy Harris, the proposed measure would adjust the definition to limit total combined THC (including all isomers and THCA) in hemp at 0.3%, replacing the current delta-9-only standard.
Moreover, any product containing a “quantifiable amount” of THC, or any similar psychoactive cannabinoid would be deemed illegal, as well as synthetic cannabinoids. An exception is carved out for true industrial hemp uses (fiber, seed, etc.), but even non-intoxicating CBD products could be swept up if they contain any detectable amount of THC.
The spending bill was approved by the House Appropriations Committee in late June. The likelihood of the provisions passing through with the spending bill remains uncertain, as similar language has appeared in prior proposed amendments to the Farm Bill, only to stall before receiving a full vote.
State Regulatory Approaches and Challenges
In the face of federal inaction, states have taken varied approaches to addressing intoxicating hemp-derived products: Some have enacted outright bans, while others have implemented regulatory frameworks with age limitations, potency caps, and licensing, to allow limited sales. These state-level efforts have been challenged by industry stakeholders under the dormant Commerce Clause and principles of federal preemption.
Although early challenges to state laws and regulations suggested that state regulatory attempts would be preempted by the 2018 Farm Bill, more recent decisions reaffirm the states’ ability to regulate these products to protect the health and welfare of their citizens.
In September 2024, the California Department of Public Health issued emergency regulations banning hemp-derived food, beverage, and dietary products with any detectable amount of THC or other intoxicating cannabinoids. The rules cover over 30 compounds, including delta-8 and delta-10 THC, and impose a prohibition on sales to individuals under 21 years old and a limit of five servings of product per package.
Industry members quickly filed suit in Superior Court for the County of Los Angeles, arguing that the agency overstepped its legal authority, failed to comply with the state’s Administrative Procedure Act, and that the regulations conflict with state and federal law.
The court in U.S. Hemp Roundtable v. California Department of Public Health rejected their arguments, holding that “[t]he Farm Bill does not prevent states from prohibiting the manufacture or storage of hemp final form food products containing detectable levels of THC. Nor does it prevent states from banning sales of these products within their borders.” The plaintiffs ultimately dropped their suit, and as a result, the regulation will remain in effect.
In Virginia, a group of hemp businesses challenged the state’s 2023 law, which capped total THC in hemp products at 0.3%, as preempted by the 2018 Farm Bill and in violation of the Dormant Commerce Clause. In January, the 4th U.S. Circuit Court of Appeals issued an opinion in which it refused to enjoin the law, rejecting the plaintiff’s preemption arguments.
Echoing the Superior Court’s reasoning in California, the 4th Circuit emphasized that “by expressly permitting states to regulate the production of hemp more stringently than federal law, the 2018 Farm Bill actually recognizes the states’ ability to regulate the production and sale of industrial hemp extracts and hemp products within their borders,” leaving states free to regulate the products under their traditional health and safety powers.
Dormant Commerce Clause attacks were likewise turned aside, as the restrictions applied equally to in-state and out-of-state products and therefore did not unduly burden out of state actors. See Northern Virginia Hemp and Agriculture v. Commonwealth of Virginia, 125 F.4th 472 (4th Cir. 2025).
More recently, the 8th U.S. Circuit Court of Appeals dissolved an injunction against Arkansas’ Act 629, which criminalizes most psychoactive hemp products by scheduling delta-8, delta-10, and other cannabinoids under the state’s Controlled Substances Act. Like Virginia, a coalition of hemp growers and retailers filed suit, claiming that Arkansas’ attempt to criminalize these products was preempted by the 2018 Farm Bill.
The court disagreed, holding that “[t]he text [of the 2018 Farm Bill] does not support [Plaintiff’s] claim that Congress intended to ‘federally protect[ ] hemp’ and coercively mandate nationwide legality,” and that “just because states may legalize hemp under the 2018 Farm Bill does not mean they must.”
Citing the Farm Bill’s anti-preemption clause (7 U.S.C. §1639p(a)(3)(A)), the court ruled that states may impose stricter hemp regulations — even outright bans — so long as interstate transport of lawful hemp is not impeded. As a result, Arkansas is now free to enforce its ban on intoxicating hemp-derived products, which state officials heralded as a major victory for public health. See Bio Gen LLC v. Sanders, (8th Cir. 2025).
Finally, also in June, the Texas Legislature passed SB 3, which would have banned all consumable hemp products that contain any amount of THC in the state. However, in a move that surprised and frustrated some lawmakers, Governor Abbott vetoed the bill before it could become effective.
Ironically, Abbott’s veto reasoning cited the now-overturned Arkansas injunction: He argued that signing a ban would mire Texas in litigation for years, as happened in Arkansas. One day later, the 8th Circuit upheld Arkansas’ law. Governor Abbott called a special legislative session and urged lawmakers to craft a regulatory framework for hemp products that is similar to alcohol, rather than an outright ban.
Why It Matters
The legal landscape for intoxicating hemp-derived products has shifted significantly over the past year. Federal efforts are underway to redefine hemp and ban synthetic and intoxicating cannabinoids. At the same time, courts have affirmed state authority to enact bans or impose strict limits. Together, courts are sending a clear message: The Farm Bill is not a shield against state bans on hemp-derived THC. As a result, states have been able to implement robust restrictions.
Businesses must stay informed and compliant as regulations continue to evolve. With federal reform looming and states tightening controls, the era of unregulated hemp-derived intoxicants may be coming to an end.
Our Cannabis Practice provides advice on issues related to applicable federal and state law. Cannabis remains an illegal controlled substance under federal law.
Troutman Pepper Locke attorneys examine the recent wave of laws and regulations targeting junk fees at both the state and federal level.
Given rising federal and state enforcement on “junk fees,” businesses should confirm their advertising reflects total prices and review consumer feedback to ensure compliance.
Junk fees refer to the additional charges on goods and services—such as convenience fees, processing fees, and service fees—that aren’t properly disclosed by businesses upfront but revealed at the final stages of a purchasing process. This tactic is also known as drip pricing.
The Federal Trade Commission late last year issued its final rule on unfair or deceptive fees, an unprecedented move to combat junk fees in the hospitality and entertainment sectors. Despite the change of executive administration, which led some to expect a potential decrease in enforcement vigor, junk fees and drip pricing practices remain a bipartisan concern.
Read the full article on Bloomberg Law.
The Bankruptcy Code provides that a debtor may voluntarily convert its case from Chapter 11 to Chapter 7 unless a Chapter 11 trustee has been appointed, the case was commenced involuntarily, or the case had previously been converted to Chapter 11.
This article will discuss the key issues of converting from Chapter 11 to Chapter 7, and how stakeholders may be affected. To access this article and read other insights from our Creditor’s Rights Toolkit, please click here.
Published in Law360 on July 17, 2025. © Copyright 2025, Portfolio Media, Inc., publisher of Law360. Reprinted here with permission.
Biotechs have faced several challenging years with slumping valuations and a competitive funding environment. However, the latest slew of retirements and layoffs at the U.S. Food and Drug Administration could present their greatest challenge yet.
While the new FDA Commissioner Martin Makary has promised speedier approvals and shorter drug development timelines, concerns persist that the agency’s reduced headcount will impede approval pathways. Companies are already reporting longer wait times for clinical trial design review and scheduling meetings with FDA personnel.[1]
These delays, by necessity, extend the timeline for drug approval, requiring biotech companies to fund operations for a longer period pending commercial product launch.
With biotech companies already pushing their cash runways to the limit, volatility in the public market and private funding at a premium, companies are turning to collaborative deal structures as an alternative source of financing and to reduce their burn rate.
Licensing and Collaborations as Alternative Sources of Financing
Out-licensing of noncore assets can provide an alternative financing option to biotech companies encountering fundraising challenges in the current market.
Typically, these deals include an up-front cash payment with additional amounts payable upon achieving certain developmental and/or commercial milestones, as well as a royalty on net sales of the resulting products. This cash can then be used to fund development of the company’s core technology.
For early-stage assets, or when the innovator biotech has specific expertise beneficial to the ongoing development of a technology, a development collaboration may be more appropriate.
Unlike an out-license, where the innovator company generally cedes control of the development process to the licensee, biotechs entering into a collaboration generally partner on the development of a product or products.
Typically, the collaboration agreement allocates responsibility for certain areas to each party and includes a requirement that the parties form a committee composed of representatives from each to oversee the entirety of the development process and to assist in decision-making and dispute resolution.
The ideal collaboration partner is one who has specific strengths — like research and development, clinical study design, manufacturing, sales force or market access — that the innovator company does not and that are needed to efficiently advance the project.
By partnering, biotechs can tap into these additional resources with no cash outlay, thereby reducing the financial burden and speeding the path to product approval, often getting a cash infusion.
In addition, biotechs without an established sales force or physician coverage for their product can leverage their collaboration partner’s network and reduce cash burn related to hiring sales staff at the commercialization stage.
In return, the collaboration partner obtains rights to market and sell the resulting product for certain therapeutic indications and/or in certain markets and geography, with the innovator retaining the balance of these rights.
Partnerships and collaborations can often lead to the acquisition of the innovator by the collaboration partner as the partner better understands the product and potential upside.
High-demand drug markets are already seeing these types of deals in action. For example, the obesity drug space is anticipated to generate more than $100 billion in revenue by 2030.[2]
In a recent move, Zealand Pharma AS entered into a collaboration with pharmaceutical giant Hoffmann-La Roche AG to commercialize an amylin analog that can be used in obesity treatment.
This $5.3 billion deal — composed of up-front and milestone payments — allowed Zealand to avoid having to sell itself, thereby preserving future upside from commercialization of the product to its shareholders.
In addition, the transaction allows Zealand to benefit from Roche’s global infrastructure and commercialization expertise. The transaction was on the tails of several other biotech licensing and collaborations in the obesity drug space in the first half of 2025.
Hart-Scott-Rodino Filings Could Cause Deal Slowdowns
One potentially countervailing factor is the impact of Hart-Scott-Rodino Act filing obligations.
The HSR Act mandates that parties exceeding certain size thresholds undertaking acquisitions over a certain size notify the government — specifically, the U.S. Department of Justice and Federal Trade Commission — in advance of closing their transaction to allow the government time to review the proposed acquisition transactions for potential anticompetitive effects.
HSR can also apply to certain patent licensing arrangements, such as those involving exclusive licenses transferring all commercially significant rights with respect to certain medical and botanical products, pharmaceutical preparations, in-vitro diagnostic substances and biological products. Commercially significant rights include grants of exclusive geographic territories or fields of use.
For these types of license transactions this year, HSR filing obligations apply if one of the parties is engaged in U.S. commerce, the parties have annual sales or assets of at least $252.9 million and $25.3 million, respectively, and the transaction is valued at more than $126.4 million.
For purposes of calculating the transaction value, the parties are required to estimate the total value of all payments over the life of the arrangement, not just the up-front payment. If the value of the transaction exceeds $505.8 million, it is reportable regardless of the size of the parties.
If the thresholds are met, both parties must file premerger notification forms and observe a 30-day waiting period before completing the transaction. The 30-day period may be extended if the government issues a request for additional information.
While many expected the current administration to sideline HSR reforms, new rules went into effect on Feb. 10, which are estimated to extend contractual filing timelines from less than 10 days to at least 30, due to more burdensome reporting requirements.
In addition, new leadership at the FTC and DOJ have indicated their intent to focus on deals in the pharmaceutical and health care industries as part of the administration’s broader efforts to lower health care costs and promote competition.
For example, the FTC has challenged the acquisition of a manufacturer of coatings for medical devices and continued its litigation against pharmacy benefits managers, while the DOJ has continued its challenge to the combination of two large home health providers.
Also, on June 11, the FTC, DOJ, U.S. Department of Health and Human Services and U.S. Department of Commerce announced three joint listening sessions on lowering Americans’ drug prices through competition, which will include panels regarding generic and biosimilar availability, prescription drug formularies and benefits and regulatory barriers.
The sessions are intended to inform the FTC and DOJ’s joint report on reducing anticompetitive behavior of pharmaceutical manufacturers, as contemplated by an April executive order.
Therefore, licensing arrangements should be assessed for HSR applicability and filing obligations, and strategies for managing competition risk should be built into any deal timeline on the front end.
Aside from the size-of-person test, two principal questions often must be examined to determine whether an HSR filing is required and a waiting period observed: whether the proposed license will transfer commercially significant rights and whether the valuation of the license exceeds the applicable size-of-transaction test.
The HSR regulations define commercially significant rights as “the exclusive rights to a patent that allow only the recipient of the exclusive patent rights to use the patent in a particular therapeutic area (or specific indication within a therapeutic area).”
The license need not transfer all the rights to make, use and sell. The governing principle is whether the licensee will receive the rights that generate profits. For example, a patent holder retaining limited manufacturing rights would still be viewed as a transfer of commercially significant rights under the HSR rules.
Although the FTC has provided guidance around this language, HSR counsel will need to understand exactly which rights will be transferred and which will remain with the patent holder, and the standard is somewhat ambiguous.
The scope of these rights frequently change during the course of the license negotiations, affecting the analysis under the HSR regulations.
Patent license valuation is also a significant question to HSR reportability and can often be difficult to determine. The acquisition price for an exclusive license is equal to the gross amount of future royalties due under the license agreement during the life of the license and milestone payments payable if certain clinical developmental targets or FDA approval are achieved.
When future royalties are too speculative to estimate reasonably, the acquisition price is considered “undetermined,” and the value of the license is the current fair market value of a fully paid-up license.
According to FTC staff guidance, the fair market value of U.S. patents should be determined using a method that is consistent with the method used for determining the value of the overall patent portfolio. The value should include any goodwill, know-how or other intangible assets allocated to those U.S. patents.
The board of the acquiring person or its designee is responsible for determining the value in good faith. The agency does not have a preferred method of valuation and does not give advice on what method is appropriate.
Beyond the HSR reporting obligation questions, any product portfolio or development pipeline overlaps between the licensee and the patentholder should be identified in order to determine the extent of the information required for the HSR filing and whether the enforcement agencies might have any concerns.
Under the new HSR rules, the time required to prepare the submission is significant, particularly because greater client interaction is necessary and typically the parties already have many other tasks to accomplish in furtherance of the licensing arrangement.
Additionally, to draft the necessary responses and locate the information required under the current rules involves more judgment calls that can only be made after counsel has a good understanding of the markets or potential markets and pipeline.
Further, this understanding is essential to decisions regarding timing and whether any antitrust risks affect the licensing agreement itself, including, for example, the appropriateness of exclusivity, pricing provisions, duration and restrictions development.
Conclusion
As biotech companies navigate FDA uncertainties and economic pressures, licensing and collaboration transactions can provide the necessary funding and pathways to advance innovative products. However, the current market and political climate will nonetheless affect deal terms and transaction considerations.
[1] https://www.law360.com/articles/2315013/fda-cuts-prompt-biotech-players-to-rethink-deal-strategies.
[2] https://www.law360.com/healthcare-authority/articles/1839686/a-changing-regulatory-landscape-for-weight-loss-drugs.
State attorneys general increasingly impact businesses in all industries. Our nationally recognized state AG team has been trusted by clients for more than 20 years to navigate their most complicated state AG investigations and enforcement actions.
State Attorneys General Monitor analyzes regulatory actions by state AGs and other state administrative agencies throughout the nation. Contributors to this newsletter and related blog include attorneys experienced in regulatory enforcement, litigation, and compliance. Also visit our State Attorneys General Monitor microsite.
Contact our State AG Team at StateAG@troutman.com.
Troutman Pepper Locke Spotlight
American Bar Association’s State and Local Government Law Section Webinar Series: State Attorneys General Enforcement Actions and Litigation: The Unwritten Rules
Join Troutman Pepper Locke attorney Ashley Taylor, the co-leader of the firm’s State Attorneys General team, as he participates in part two of the American Bar Association’s State and Local Government Law Section webinar series titled “State Attorneys General Enforcement Actions and Litigation: The Unwritten Rules.” This session will focus on the “Multistate Investigations and Settlements” chapter from the recently published book, Consumer Protection: Understanding Enforcement Actions Brought by State Attorneys General. The webinar aims to delve into the complexities and nuances of enforcement actions initiated by consumer protection staff within state attorneys general offices.
State AG News
New York AG Intensifies Investigation and Enforcement Against Alleged Medicaid Fraud
By Troutman Pepper Locke State Attorneys General Team and Philip Nickerson
On June 30, 2025, New York Attorney General (AG) Letitia James announced new lawsuits, criminal charges, and settlements with 25 New York transportation companies related to alleged schemes to defraud Medicaid of millions.
Newly Appointed Wyoming AG, Keith Kautz
By Troutman Pepper Locke State Attorneys General Team
Keith Kautz will serve as the new attorney general (AG) of the state of Wyoming following his appointment by Governor Mark Gordon last week, effective July 7, 2025. Kautz has a long history of civil service in the state, having served as a district court judge for the Eighth Judicial District — covering Converse, Goshen, Niobrara, and Platte counties — for 22 years. His hometown of Torrington falls within this district. In 2015, Governor Matt Mead appointed Kautz as a justice to the Wyoming Supreme Court, where he served until his retirement in March 2024.
AG of the Week
Brian Schwalb, District of Columbia
Brian L. Schwalb is the District of Columbia’s second independently elected attorney general (AG). He is a third-generation resident of Washington, D.C. Schwalb completed his undergraduate studies at Duke University and earned his law degree from Harvard Law School. Following his education, he undertook a two-year judicial clerkship and subsequently worked as a trial attorney at the U.S. Department of Justice. After his tenure at the Justice Department, Schwalb transitioned to private practice, where he represented a diverse range of clients, including individuals, businesses, nonprofits, and families, in various high-stakes legal matters. His work included advocating for individuals affected by excessive police force, those defrauded of their assets, and individuals on death row.
Prior to his election as AG, Schwalb held leadership roles in the legal field, serving as Venable’s firmwide vice chairman and as the partner-in-charge of Venable’s Washington, D.C. office. He is recognized professionally as a fellow of the American College of Trial Lawyers.
District of Columbia AG in the News:
-
On July 15, Schwalb announced a settlement with pharmaceutical company Gilead Sciences, Inc., resolving allegations related to illegal kickbacks.
-
On July 8, Schwalb announced two settlements with related cleaning companies to resolve allegations related to unpaid overtime wages.
-
On June 24, Schwalb announced three lawsuits against Maryland drivers, and one against a Virginia driver, alleging the four individuals had amassed more than 1,000 traffic infractions among them.
Upcoming AG Events
-
July: DAGA | Presidential Partners Retreat | Santa Fe, NM
-
August: AGA | Chair’s Initiative | Alaska
-
September: RAGA | Fall National Meeting | Miami, FL
For more on upcoming AG Events, click here.
The U.S. Department of Justice (DOJ) has announced that it is expanding its enforcement priorities to include a focus on import-related fraud — particularly schemes aimed at evading U.S. tariffs and duties. This marks a notable shift in the DOJ’s trade enforcement agenda, which has traditionally emphasized export controls and sanctions compliance.
The shift follows a newly issued memorandum by the DOJ’s Criminal Division, outlining how tariff evasion will be treated as a central enforcement priority moving forward. Companies that import goods into the U.S. — or rely on suppliers who do — should be aware that trade fraud involving customs declarations is now firmly within the DOJ’s crosshairs.
What’s Changing?
The DOJ is now devoting significant investigative and prosecutorial resources to conduct that undermines U.S. customs laws. This includes:
-
Undervaluing imported goods to reduce duties owed;
-
Falsifying country-of-origin information, including deceptive labeling or transshipment to conceal origin;
-
Misusing free trade agreement preferences (e.g., under the U.S.-Mexico-Canada Agreement) without meeting eligibility requirements;
-
Improperly classifying products to secure a lower duty rate or avoid tariffs entirely; and
-
Structuring transactions to sidestep tariffs, such as Section 301 duties on Chinese-origin goods or Section 232 tariffs on steel, aluminum, automobiles, and automotive parts.
Expansion of Trade Enforcement
Historically, tariff enforcement has been the domain of U.S. Customs and Border Protection (CBP) and civil penalty proceedings. The DOJ’s involvement adds a criminal dimension that significantly raises the stakes for businesses and individuals involved in international trade.
This policy shift also aligns with recent trends: government agencies are increasingly sharing trade data, using advanced analytics to flag anomalies, and coordinating multiagency investigations. The DOJ has indicated it will work closely with CBP, Homeland Security Investigations, and the Department of Commerce to identify and prosecute trade fraud schemes.
As discussed in our prior client alert, the DOJ is increasingly applying the False Claims Act to customs-related violations, signaling a broader enforcement approach. One recent example is the case of United States v. Barco Uniforms Inc., where the government alleged that the importer falsely identified the origin of its goods in order to reduce or avoid applying applicable tariffs. The matter reflects DOJ’s growing use of civil fraud statutes — alongside traditional criminal charges — to target inaccurate or misleading trade-related disclosures and recover lost government revenue. Furthermore, in light of the DOJ’s recent expansion of its Corporate Enforcement Policy — specifically extending the Criminal Division’s whistleblower pilot program to cover tariff and customs fraud — both companies and individuals should anticipate a rise in investigations and potential enforcement actions in this space.
On July 10, the DOJ announced it is reallocating prosecutorial and investigative resources to support its expanded focus on customs and tariff enforcement. Notably, the DOJ Criminal Division will refocus the Market Integrity and Major Frauds Unit — which has historically handled large-scale securities and commodities fraud — to prioritize tariff evasion schemes. The unit will be renamed the Market, Government, and Consumer Fraud Unit, and will be bolstered by personnel from the DOJ’s Civil Division, including the Consumer Protection Branch.
What Companies Should Do Now
In light of this expanded enforcement posture, companies should revisit their import compliance frameworks with a focus on risk exposure and internal accountability. Key action items include:
-
Auditing import records and customs filings for potential misstatements or misclassifications;
-
Reviewing supplier declarations and country-of-origin certifications, especially for goods sourced from high-risk jurisdictions;
-
Ensuring trade compliance policies and training are updated and applied consistently across the business;
-
Conducting internal investigations where potential red flags exist and preparing for voluntary disclosure where appropriate.
Failure to act could expose companies not only to monetary penalties but also to reputational damage, criminal liability, and disruption of supply chains.
Looking Ahead
The DOJ’s decision to treat customs fraud as a criminal enforcement priority represents a material development in the trade compliance landscape. For companies operating in sectors affected by tariffs, trade remedy duties, or complex origin rules, proactive compliance is no longer optional — it is essential.
This is intended as a guide only and is not a substitute for specific legal or tax advice. Please reach out to the authors for any specific questions. We expect to continue to monitor the topics addressed in this alert and provide future client updates when useful.





